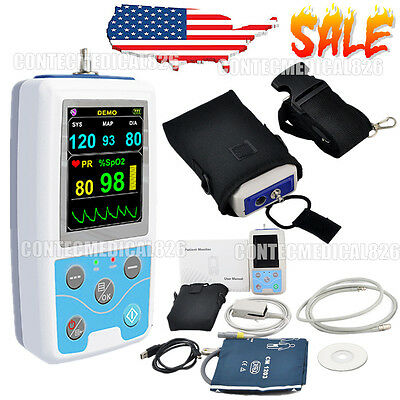
Vital Signs Monitor Patient Monitor SPO2,NIBP,Pulse Rate,24hrs Ambulatory NIBP
$78.67 (-40%)
PM50 Patient Monitor,24 Hours recorder/analyzer,PC Software,SPO2+NIBP+PR
Introduction
PM50 is a multi-functional patient monitor
that can monitor BP and SpO2 at the same time. Realizing long time monitoring of
dynamic blood pressure, the device is widely applicable to hospital wards,
community clinics and other medical institutions.
Functions
Features:
1)Compact and portable, easy
to use.
2)With Integrative SpO2 Probe.
3)Double working modes,monitoring
function and 24 hours ambulatory NIBP measurement function can be flexibly
set.
4)Long time monitoring BP and SpO2, displaying value of BP, SpO2 and
PR.
5)NIBP and SpO2 data Record for large capacity.
6)With friendly user
interface, the user can see list menu and review measurement results.
7)The
device can display low power information, alarm information, error information
and time information richly.
8)Parameter alarms function is
optional.
9)Patient information can be flexibly set.
10)Case management
function.PC software can achieve data review, analysis measurement results,
seeing trend, printing reports and other functions.
11)Data can be
transmitted between device and computer, such as upload patient information,
download measurement data.
12)English and Chinese menu.
Function of master
device software:
1)Set patient information through PC software.
2)Up to
1000 patients cases can be edited and supervised.
3)The time segment of
dealing with the patient’s NIBP data is 48 hours.
4)Connect the device by USB
interface.
5)Can upload patient information data collection project and
download collection data.
6)Can display scoop-shape trend graph,filling-type
trend graph, histogram, pie chart, correlation line graph.
7)Can edit every
piece of data, and add annotation to it.
8)Can edit basic
information,doctor’s advice information, NIBP and SpO2 status instruction,
current medicine-taken information, etc.
9)Support print preview,print the
report.
Performance
NIBP:
Measurement Method:
Oscillometry
Measurement Mode: The upper arm measure
Measuring range:
SYS 40mmHg~270mmHg
MAP 20mmHg~235mmHg
DIA
10mmHg~215mmHg
Automatic Measurement Interval: 5.10.15.20.30.45.60.90.120
minutes
Resolution: 1mmHg
Accuracy: ±3mmHg
Increasing pressure mode:
force pump increases pressure automatically
Reducing pressure mode:
self-motion ladder reducing pressure mode
Alarm parameter: SYS /
DIA
SpO2:
Measuring range: 0%~100%
Resolution:1%
Accuracy:
70-100%, ±2%, below 70% unspecified
Error in Weak Filling Condition:SpO2 and
pulse rate can be shown correctly when pulse-filling ratio is 0.4%. SpO2 error
is ±4%, pulse rate error is ±2 bpm or ±2% (select larger).
Resistance to
surrounding light:The deviation between the value measured in the condition of
man-made light or indoor natural light and that of darkroom is less than
±1%.
Pulse Rate:
Measuring
range:30bpm~250bpm
Resolution:1bpm
Accuracy: ±2 bpm or ±2% (select larger)
Display:
2.4″ TFT colour LCD
Safety:
Power: DC 3V(Two “AA”,1.5V
Alkali Battery)
Product safety type:Type BF application part (Internally
powered,defibrillator protected)
Accessories
cuff for adult
Integrative SpO2
Probe
USB data line
Disk (PC software)
Pouch
User manual
Physical
characteristic
Dimension: 128*69*36 mm (No including
Packing)
Weight: <350g (Including Batteries)
Working
environment:
Temperature:5 ˚C~40 ˚C
Relative
humidity:≤80%
Altitude:-500 to 4,600m (-1,600 to 15,000ft)
Specific EMC,
climate, mechanical environment: Do not use a mobile phone in the vicinity of
the patient monitor for the strong radiation field produced by mobile phone
would interfere with the normal function of the patient monitor use.
Storage
environment:
Temperature: -20˚C ~ 60 ˚C
Relative humidity: ≤95
%
Altitude:-500 to 13,100m (-1,600 to 43,000ft)
Specific EMC, climate,
mechanical environment: The patient monitor after packaged should be stored in a
well-ventilated room with no corrosive gas. Strong shock, vibration and snow and
rain should be avoided during transportation.
Buy safe Products
The following FDA Disclaimer is required for all eBay listing in Healthcare category and is included for REFERENCE:
The sale of this item may be subject to regulation by the U.S. Food and Drug Administration and state and local regulatory agencies.If the item is subject to FDA regulation,we will verify your status as an authorized purchaser of this item before shipping of the item.All of our products are CE approved.
If you have questions about legal obligations regarding sales of medical devices, you should consult with the FDA’s Center for Devices and Radiological Health.
The Fingertip Pulse Oximeter is registered on the Australian Register of Therapeutic Goods (ARTG) with the code 197923, and certified by FDA of United States and CE,TUV of Europe.The Fingertip Pulse Oximeter that is FDA 510K Approved
Terms of Sale
There are not extra taxes,VAT or other unmentioned payment.You pay us with goods subtotal + shipping cost (not include
duties). But you need to pay for the import duties in case that occurred for certain goods, or you may kindly give us some
suggestions about that, we promise to do our best to help you!
International Buyers – Please Note:
Import duties,taxes and charges are not included in the item price or shippingcharges.These charges are the buyer’s
responsibility.Please check with your country’s customs office to determine what these additional costs will be prior to
bidding/buying.”
Contact US
Contec Medical Systems—Tessy
Skype:contec.tessy
Email:[email protected]
Whatsapp/Wechat/Mobile:86-18716007715
Only English user guide, if you need any other language, please contact us!
Only English user guide, if you need any other language, please contact us!